FAQ
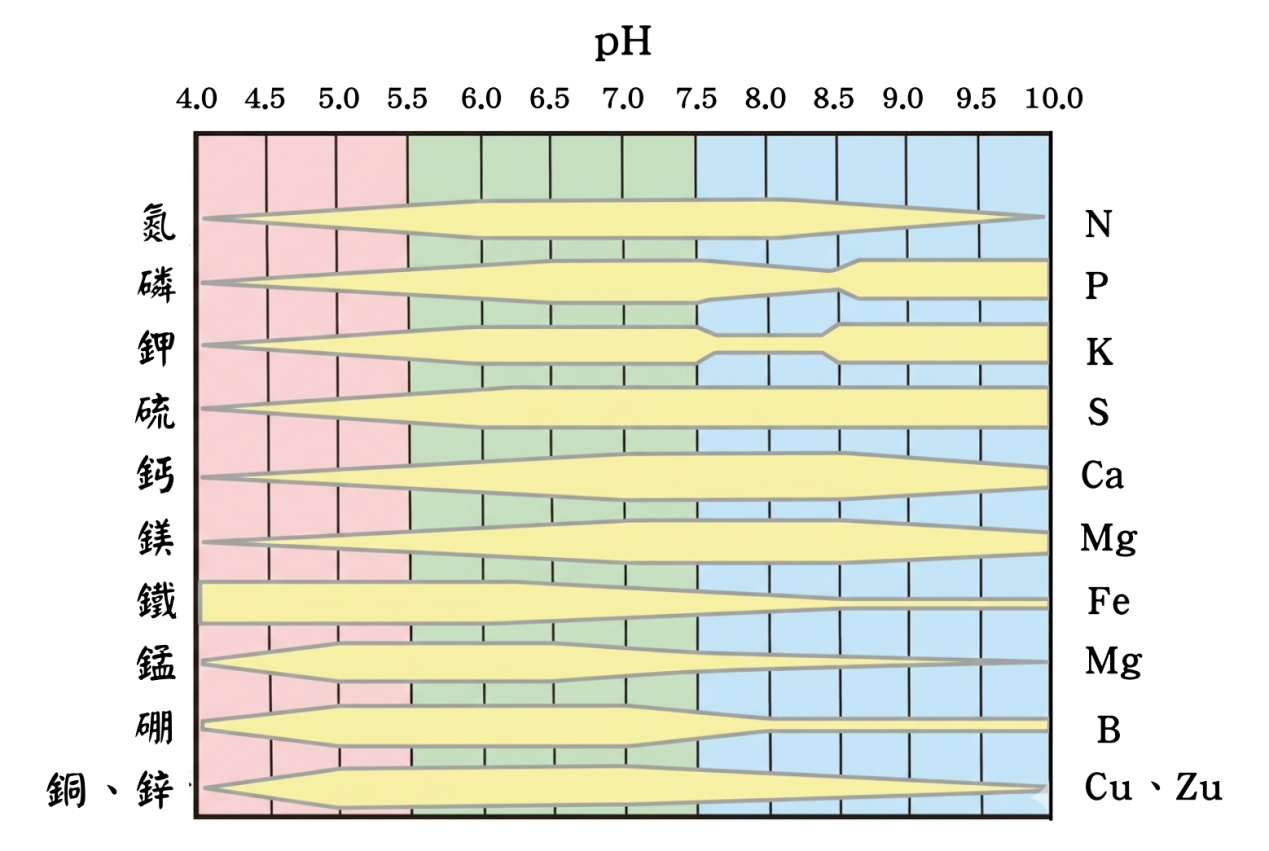
Soil pH is a key indicator used to measure soil acidity and alkalinity, and it directly affects plant growth and nutrient uptake. Most crops grow best within a pH range of 5.5 to 7.5, where nutrient availability is at its highest. When soil becomes too acidic, elements such as aluminum and iron are more likely to dissolve, potentially causing toxicity to plants. At the same time, essential nutrients like phosphorus, calcium, and magnesium may become fixed in the soil, reducing their availability for plant absorption.
On the other hand, if the soil is too alkaline, deficiencies of micronutrients such as iron, zinc, and manganese may occur, leading to physiological disorders like chlorosis.
In addition, soil pH also influences the activity and composition of soil microorganisms. Most beneficial microbes prefer neutral to slightly acidic conditions. If the pH is unsuitable, it can reduce the efficiency of organic matter decomposition and nutrient cycling, thereby affecting overall soil fertility. Therefore, adjusting soil pH through appropriate materials can improve the growing environment and enhance crop yield and quality.
Back
